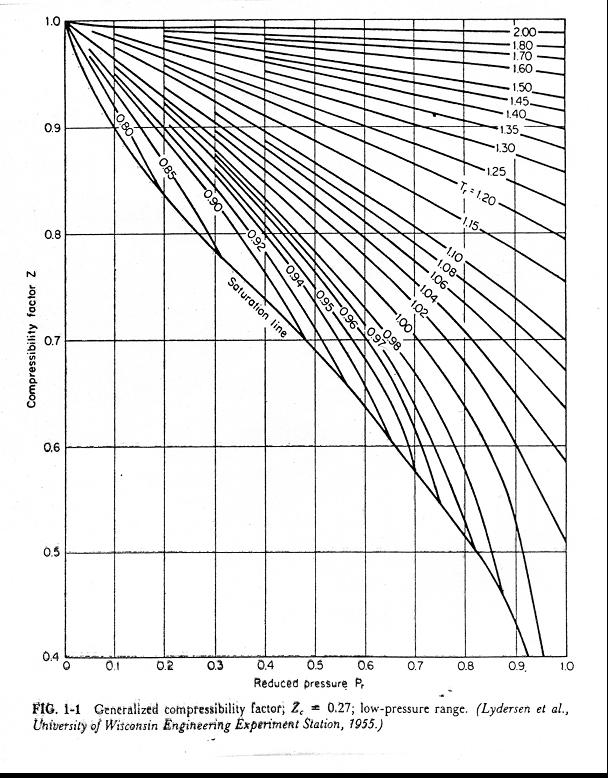
OneClass: For a real gas, the compressibility factor, Z, is
5 (495) · $ 20.99 · In stock

The compressibility factor for a real gas is expressed by, z =1+ BP / RT. The value of B at 500 K and 600 bar is 0.0169 L / mol. Find the
If z<1, does it mean that the gases behave more like perfect or real gases? - Quora

What is compressibility factor? What is its value for ideal gas

OneClass: For a real gas, the compressibility factor, Z, is defined as Z (T, P) = PV/nRT For an ideal
Ideal gases and real gases are compressible or not compressible what is the compressible factor for real gases and ideal gases.

Compressibility Factor Z Important Concepts and Tips for JEE Main

Solved Real gas effects can be expressed as departures from

Compressibility factor (gases) - Citizendium

Gas Compressibility - an overview

The compressibility factor `(Z=PV//nRT)` for `N_(2)` at `223 K` and `81.06 MPa` is `1
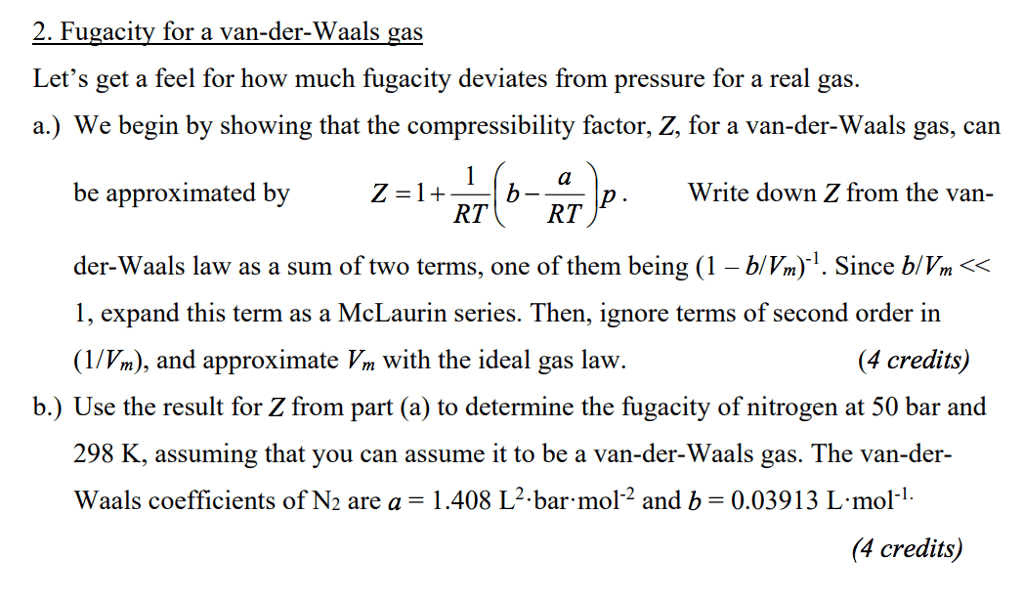
OneClass: At low pressures the compressibility factor for a Van der Waal's gas is given by Z-1+[b- (a

Real gasses For an ideal gas, the compressibility factor Z = PV/nRT is equal to unity for all conditions. For a real gas, Z can be expressed as a function. - ppt