Compression Factor Z
4.8 (345) · $ 31.00 · In stock

The Compression Factor, Z, and Real Gases - What you NEED to Know

3.2 Real gas and compressibility factor – Introduction to

Solved The compression factor, Z, can also be calculated

e Compressibility factor (Z) for hydrogen WRT pressure and
The value of compression factor at the critical state of a vander

The compression factor (Z) Co, 7°C and 100 atm is 0.21. Calculate the volume of a 4 mole sample of co, same temperature and pressure (use R = 0.08 L. atm/K.mol (1)

Solved (Triple-Play Bonus) For a certain gas, the
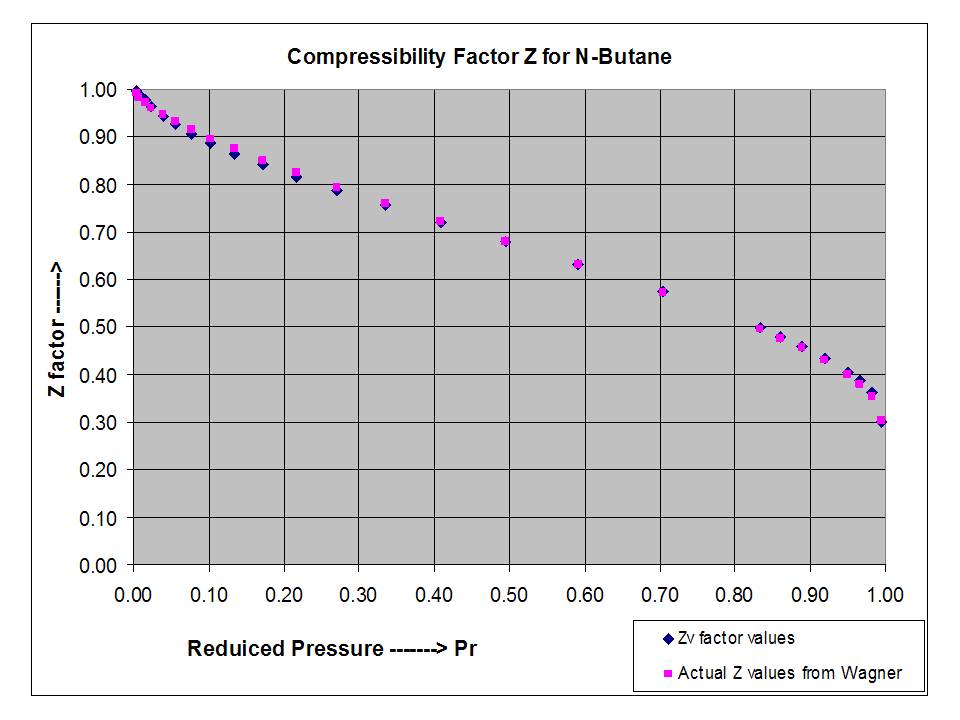
Two extensions of the compressibility factor Z correlation (sub

physical chemistry - Is the compressibility factor smaller or greater than 1 at low temperature and high pressure? - Chemistry Stack Exchange
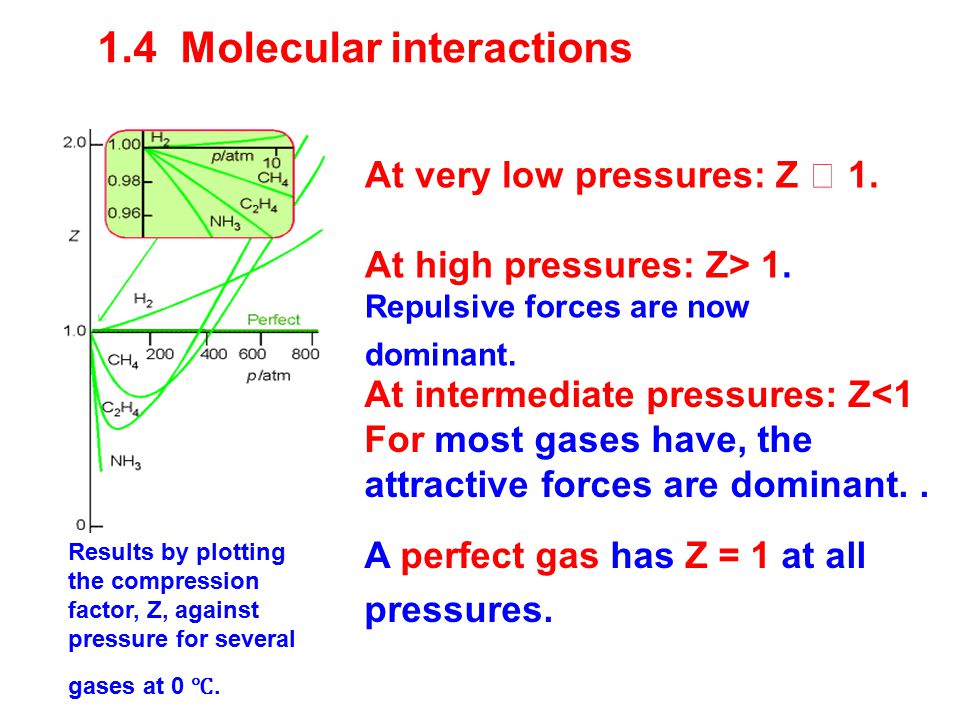
Real gases 1.4 Molecular interactions 1.5 The van de Waals equation 1.6 The principle of corresponding states Real gases do not obey the perfect gas law. - ppt download

Answered: (a)Using the compressibility chart,…